Overview of the management of bronchiectasis (see below for the management of exacerbations)
The management of bronchiectasis is not strongly supported by clinical trial evidence and clinical practice guidelines have largely been formulated through consensus in expert opinion. However, the majority of patients with bronchiectasis produce sputum on a daily basis so it is clinically accepted that the clearance of secretions from the airways should be the cornerstone of daily treatment.
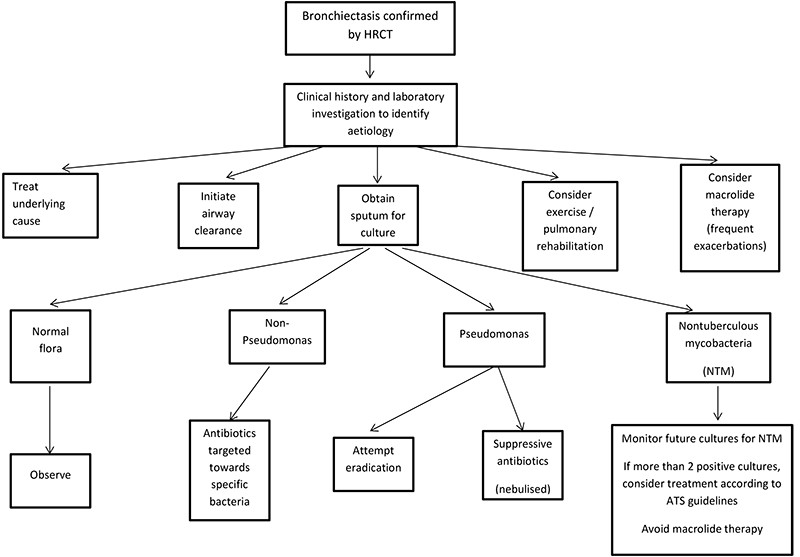
The development of a management plan (Fig. 1) for patients with bronchiectasis should be guided by the input of a respiratory physician and a physiotherapist with experience in airway clearance techniques. The constituents of the management plan will be guided by symptom burden, exacerbation frequency, sputum microbiology, lung function and the presence of exacerbating factors, respiratory comorbidity (asthma, COPD etc) and disease complications.
In general, the aim is to optimise well-being, symptom control, lung function, quality of life (QoL), and reduce exacerbation frequency and prevent excessive decline in lung function.
Treatment plans for exacerbations should be developed for all patients, linking them to primary healthcare and specialist or hospital facilities. When appropriate, this includes individualised and self-initiated management action plans.
Airway clearance manoeuvres and regular exercise are encouraged, nutrition optimised, environmental pollutants (including tobacco smoke) avoided and vaccines administered following national immunisation schedules. Long-term antibiotics, inhaled corticosteroids, bronchodilators and mucoactive agents may be individualised, but are not recommended routine therapy.
Long-term oral or nebulised antibiotics should not be used routinely and the development of antibiotic treatment plans should be based on specific patient characteristics guided by microbiology results and expert advice.
Antibiotic selection is based upon lower airway culture results, local antibiotic susceptibility patterns, clinical severity and patient tolerance. Patients with severe exacerbations and/or not responding to outpatient therapy are hospitalised for more intensive treatments, including intravenous antibiotics.
Inhaled and oral corticosteroids should not be prescribed routinely unless there is an established diagnosis of co-existing asthma or COPD. Similarly, inhaled bronchodilators should not be prescribed routinely and used only on an individual basis.
Mucoactive agents, including saline, hypertonic saline and mannitol, are not currently recommended for routine use. A therapeutic trial however may be considered in children and adults with frequent exacerbations.
For further guidelines see: European Lung Foundation – managing bronchiectasis in adults
Lung Foundation Australia: LFA Bronchiectasis Stepwise Management Plan
See TSANZ Bronchiectasis Guidelines
Treatment for bronchiectasis aims to:
Treatment for bronchiectasis aims to:
Decrease symptoms (such as cough and fatigue)
Decrease frequency and severity of exacerbations
Decrease hospital admissions
Improve quality of life
Improve exercise tolerance
Maintain or improve lung function
Prolong survival
Defining patient specific treatment goals.
Identifying
Symptoms type and duration
Sputum volume and characteristics (colour thickness, smell, ease of clearance, presence of blood)
Frequency of exacerbations (previous hospitalisations and antibiotics)
Responses (and adverse effects) to previous treatments (antibiotics)
Haemoptysis volume and frequency
Identifying co-morbidities that can contribute to bronchiectasis symptoms
Sinusitis
Gastro Oesophageal Reflux (GORD)
Asthma / ABPA
COPD and chronic bronchitis
Identify and promoting self-care activities
Exercise
Airway clearance
Symptom monitoring
Management of acute exacerbations
Antibiotic Therapy
Duration of therapy
Indications for Hospitalisation
If a patient requires intravenous antibiotics, hospitalisation is indicated. For patients who have Pseudomonas aeruginosa resistant to ciprofloxacin, intravenous therapy is indicated. Other indications for hospitalisation include the presence of respiratory failure with a respiratory rate >/= 25/min, hypotension, fever >38C, hypoxic respiratory failure with SpO2 </= 92% or failure to improve after a course of oral antibiotics. It is important that patients admitted to hospital are also treated with appropriate airway clearance techniques.
There is little evidence to support or oppose the long-term use of oxygen therapy to reduce mortality in patients with chronic respiratory conditions other than COPD (TSANZ Oxygen Guidelines)
Supplemental oxygen therapy may be used if there is evidence of hypoxic respiratory failure (SpO2 < 90% or PaO2 < 65mmHg). While this may improve oxygenation, it will not necessarily have any impact on dyspnoea. If supplemental oxygen is used, it is appropriate to maintain a saturation of >92%.
The addition of oxygen therapy to a patient’s management plan should be determined by a comprehensive clinical assessment, not just the requirement of an increase in PaO2.
For further information on oxygen therapy see TSANZ Oxygen Guidelines
