Nontuberculous mycobacteria (NTM)
NTM are important environmental pathogens that have been recognised as a cause of pulmonary infection for many years, cause significant morbidity and mortality and can infect patients with pre-existent lung diseases, particularly bronchiectasis.
Infection is often associated with increased pulmonary and systemic symptoms and decline in lung function. NTM can also infect at risk individuals and be associated with the development of bronchiectasis, small airway mucous plugging and nodules.
As NTM organisms are commonly occurring microbes in the environment (particularly soil and water), patients are regularly exposed and screening should occur at regular intervals. Clinicians need to be astute in assessing the significance of NTM isolation in the individual patient, and guidelines exist to help in this process.
What are NTM?
Nontuberculous mycobacteria (NTM) consist of >180 species of mycobacteria other than M. tuberculosis complex and M. leprae. They have been divided into two categories:
those that grow in culture within 7-10 days (RGM: rapid growing mycobacteria, e.g abscessus, M. fortuitum)
those that take longer (SGM: slow growing mycobacteria, e.g avium complex, M. kansasii)
The predominant source of NTM organisms is still assumed to be the environment, with evidence implicating dust, soil, water and shower aerosols in patient acquisition of infection.
Diagnosis
Due to the presence of organisms in the environment and the wide variety of clinical manifestations, diagnosis relies on a constellation of clinical features, radiographic findings and microbiological studies.
The minimum evaluation of a patient with suspect NTM-PD should include the following:
Clinical and radiographic investigations (both required):
Pulmonary symptoms, nodular or cavity opacities on chest radiograph, or a HRCT scan that shows multifocal bronchiectasis, alongside multiple small nodules
AND
Appropriate exclusion of other diagnoses, especially tuberculosis and mycosis
Microbiological investigations:
Positive culture results from at least two separately expectorated sputum samples. If results are non-diagnostic, consider repeat smears and cultures
OR
OR
Trans-bronchial or other lung biopsy with mycobacterial histopathological features (granulomatous inflammation or stainable acid-fast bacilli), and positive culture for NTM, or biopsy showing mycobacterial histological features, and at least one sputum or bronchial washing that is culture positive for NTM
More recent guidelines published by the United States Cystic Fibrosis and European Cystic Fibrosis Society (USCF & ECFS) and the British Thoracic Society (BTS) additionally outline a suggested algorithm as follows:
Expectorated sputum, induced sputum, bronchial washings/lavage (BAL), or transbronchial lung biopsy (TBLBx) can all be used for diagnosis, with the least invasive option in the first instance
Processing of samples should occur within 24 hours of collection
Samples should be cultured on both liquid and solid media for a minimum of 8 weeks (up to 12 if necessary)
NTM isolates should be identified to the species level (except for MAC), and to the sub-species level for abscessus, using validated molecular or mass spectrometry techniques
NTM isolates should be typed with whole genome sequencing (WGS) when person-person transmission is suspected
DST should be done for MAC (clarithromycin and amikacin); kansasii (rifampicin only); and M. abscessus (at least clarithromycin, cefoxitin and amikacin; and preferably tigecycline, imipenem, minocycline, doxycycline, moxifloxacin, linezolid, co-trimoxazole and clofazimine)
CXR and HRCT should be performed in all patients with suspected NTM-PD with supporting imaging consisting of centrilobular nodules, tree-in-bud, bronchiectasis (particularly in the right middle lobe and lingula), consolidation and cavitation
Speciation and molecular techniques
Although non-culture based methods are not currently recommended for the initial detection of NTM respiratory isolates, positive isolates should undergo molecular testing for correct identification to the species level, and to the subspecies level for M. abscessus complex. There is no current gold standard molecular technique that is recommended for speciation of NTM isolates.
Common tests currently utilised include line probe assays (i.e. Hain CM and AS), PCR product restriction analysis, and partial gene sequencing (i.e. 16S, hsp65, rpoB, 16S-23S ITS) and matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry.
Drug susceptibility testing
Drug susceptibility testing (DST) for NTM should follow CLSI guidelines however it should be emphasized that its clinical value remains mostly uncertain with poor correlation between in-vitro susceptibility and in-vivo response (with the exception of macrolides in MAC-PD). It is accepted practice that DST should occur as follows:
mycobacterium avium complex (MAC) – clarithromycin and possibly amikacin
kansasii – rifampicin
abscessus – at least clarithromycin, cefoxitin and amikacin (and preferably tigecyline, imipenem, minocycline, doxycycline, moxifloxacin, linezolid, co-trimoxazole and clofazimine)
Other investigations
There are a number of other investigations that should be considered during the diagnostic work up of individuals with suspect or proven NTM-PD. Although the risk of NTM infection associated with CF (i.e. homozygous CFTR mutations) is well established, there are increasing numbers of reported CFTR polymorphisms, that predispose to bronchiectasis and NTM-PD. Moreover, rates of heterozygosity for CFTR mutations within the non-CF population with NTM-PD are reported between 30-50%. As targeted therapy evolves, specific testing for CFTR mutations should be considered in the appropriate clinical setting.
Additionally, as outlined above, specific immune deficiencies are a risk factor for NTM infection. In cases of persistent or recurrent severe infections, or in disseminated disease, further immunological investigations should be considered. Specific tests that may be ordered include lymphocyte subsets, immunoglobulin levels including IgG subclasses, HIV serology, anti-IFNg and IL-12 antibodies, and cytokine release assays.
Epidemiological studies have demonstrated an association between vitamin D deficiency and infection with both TB and NTM, in addition to in vitro treatment studies showing that vitamin D plays a critical role in host defense against mycobacteria. Thus, investigating for vitamin D deficiency, with supplementation if required, is reasonable.
Management
Diagnosing pulmonary NTM infection/disease does not equate to the need for immediate treatment. Treatment constitutes an important undertaking for the physician and patient as it usually involves combination antibiotic therapy for 12–24 months. In some cases disease remits spontaneously. Indeed, lung involvement may range in severity from subclinical/ mild clinical (indolent) infection to disease associated with extensive invasion/destruction of the lungs.
Early studies of MAC disease demonstrated a variable progression of infection, but the majority of symptomatic (and some supposedly asymptomatic) patients had progressive disease, and often died as a result.
For further information on the management of people with NTM go to: When and how to treat pulmonary non-tuberculous mycobacterial diseases
combination antibiotic therapy for 12–24 months. In some cases disease remits spontaneously. Indeed, lung involvement may range in severity from subclinical/ mild clinical (indolent) infection to disease associated with extensive invasion/destruction of the lungs.
Early studies of MAC disease demonstrated a variable progression of infection, but the majority of symptomatic (and some supposedly asymptomatic) patients had progressive disease, and often died as a result.
For further information on the management of people with NTM go to: When and how to treat pulmonary non-tuberculous mycobacterial diseases
Summary of management principles:
Important to establish that the patient has disease
Patients respond better if treated according to the guidelines
Microbiological surveillance for response to treatment and then for re-infection
Clinical monitoring for side effects
Adjunctive therapies – airway clearance and nutrition
Awareness of being a lifetime disease
Mycobacterium Avium Complex (MAC) – treatment
| ATS (2007) Initial treatment of Nodular BE | ATS Initial treatment of Cavitary disease | ATS Advanced (severe) or pretreated disease | BTS (2017) (thrice weekly or daily based on disease severity) | JTS (2013) | |
|---|---|---|---|---|---|
| Macrolide | Clar 1000mg TIW Or Azi 500-600mg TIW | Clar 500-1000mg/d Or Azi 250†-300mg/d | Clar 500†-1000mg/d Or Azi 250†-300mg/d | Azi or clari | Clar 15-20mg/kg/d 600-800mg/d |
| Ethambutol | 25mg/kg/d TIW | 15 mg/kg/d | 15 mg/kg/d | 15 mg/kg/d | 15mg/kg/d (max 750mg/d) |
| Rifamycin | Rifampicin 600mg TIW | Rifampicin 450† -600mg/d | Rifabutin 250-300mg/d or Rifampicin 450†-600mg/d | Rifampicin 450† -600mg/d | Rifampicin 10mg/kg/d (max 600mg/d) |
| Aminoglycoside | None | None or Amikacin or Streptomycin | Amikacin or Streptomycin | Amikacin/streptomycin in severe disease or treatment failure | Streptomycin or kanamycin (each <15mg/kg 2-3x/wk, max 1000mg) |
| Other drugs | Isoniazid 300mg/d or Ciprofloxacin 750mg bd | ||||
| Duration | 12 mths culture neg | 12 mths culture neg | 12 mths culture neg | 12 mths culture neg | 2 years |
In macrolide resistant MAC mortality is high without surgery and IV aminoglycosides, and similar to MDR-TB
In the early monotherapy studies of macrolides in MAC, approximately 15% of patients developed macrolide resistance by 3 months.
The two most important risk factors are:
inappropriate first line therapy – including macrolide monotherapy
dual therapy with a macrolide and fluoroquinolone
The inclusion of ethambutol in the regimen appears critically important for preventing emergence of macrolide resistance
Patients with macrolide resistant MAC should be managed in centres’ with expertise in NTM management as surgery and prolonged intravenous aminoglycosides are usually required for optimum outcomes.
M. abscessus -taxonomy (Fig. 1)
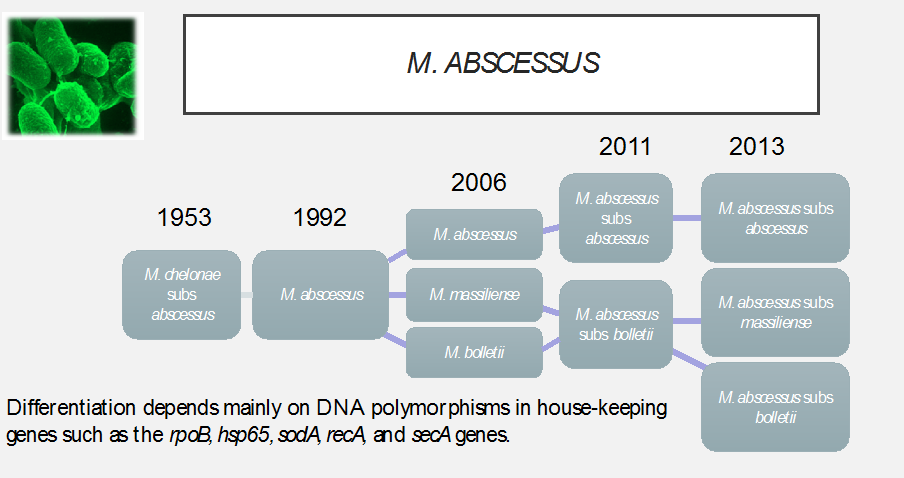
The expression of a functional erm gene in M. abscessus confers inducible resistance to macrolide antibiotics. This is determined by prolonged incubation (14 day) susceptibility testing of isolates.
Subspeciation of M. abscessus isolates generally reveals that approximately 80-100% of subs abscessus and bolletii have function erm genes, whereas M. abscessus subs massiliense isolates contain a mutation that renders the gene non-functional and the isolates susceptible to macrolides. This has been associated with significant differences in treatment success rates in published studies.
Treatment of M. abscessus infections requires a combination of intravenous and oral agents. The most current guidelines are those published by the BTS NTM Guideline – Summary of Recommendations and US and European Cystic Fibrosis Foundation.
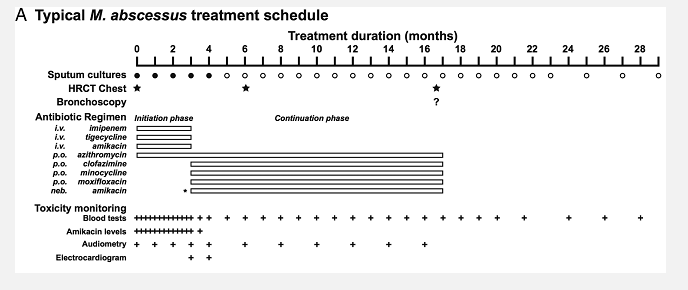
As these infections are uncommon, there is no high level evidence to support treatment options. The NHMRC-MRFF funded clinical trial of M. abscessus infections is currently evaluating the guideline based regimen against newer treatment combinations, and clinicians are urged to refer to one of the centres involved in this study.
For other species of NTM not covered by the guidelines, expert advice is recommended.
Surgery
Patients with localised disease – especially upper lobe cavity
Patients in whom drugs fail to convert the sputum cultures to negative after six months of continuous treatment
Patients who cannot tolerate the necessary drugs
Massive haemoptysis
Extensive disease – excision of large cavity foci may assist medical management of remaining lesions
Monitoring of patients with suspected or definite NTM disease
Patient whose NTM isolate is unlikely clinically significant:
May suffice to follow up every 3-6 months with clinical review, sputum and radiology (CXR and/or HRCT) until confident there has been exclusion of disease
Patients with possible or likely clinically significant isolates whom are not on treatment:
Close monitoring for symptoms, sputum, and radiology should occur at least every 3-6 months for up to 24 months or greater, until disease is confidently established or excluded
Patients who have an isolate that is definitely clinically significant and on treatment:
A suggested follow up program is as follows:
monitoring of clinical symptoms and drug toxicities every 4-8 weeks
collection of three expectorated or induced sputum for AFB and culture every 4- 8 weeks
radiology (CXR and/or HRCT) every 6-12 months
For individuals who cannot provide an adequate induced or expectorated sputum, a HRCT should be performed every 12 months with targeted bronchoscopy (wash/BAL) if required.
